Background: Recent meta-analytic evidence indicates that survivorship care plans (SCPs) may not yield improvements in cancer survivors’ patient-reported outcomes.
Objective: We sought to investigate the prevalence of SCP receipt and explore associations between SCPs and patient-reported psychosocial and health behavior outcomes in a sample of cancer survivors enriched with individuals with a family history of cancer.
Methods: Using a cross-sectional survey, we evaluated receipt of SCPs and/or treatment summaries and psychosocial and behavioral health outcomes in 123 survivors of cancer.
Results: Most (93%) of the sample had a family history of cancer; only 31% (n = 38) received some form of written care plan (SCP/treatment summary). Receipt of a care plan was associated with significantly lower psychological distress (F [1, 104] = 8.316, P = .005) and higher coping self-efficacy (F [1, 104] = 6.627, P = .011), but it was unrelated to patient satisfaction, quality of life, cancer-related distress, and other health behaviors.
Discussion: Results suggest that SCPs are infrequently provided and not uniformly implemented, even among high-risk patients. Similar to a recent systematic review, findings provide initial support for the association between written care plans and psychological distress and coping self-efficacy. Although these constructs have implications for cancer survivors’ communication with providers, psychosocial adjustment, and seeking/understanding medical information, attention to how high-risk survivors respond to SCPs and adhere to recommended follow-up care is important.
Conclusion: Results of the current study contribute to the growing literature about SCPs. Future work will need to identify whether high-risk cancer survivors benefit from SCPs over time.
Nearly 17 million cancer survivors live in the United States. Estimates suggest this number will grow to >21 million over the next decade.1-4 For survivors, late effects of cancer and its treatment include osteoporosis, hypothyroidism, cognitive problems, and secondary cancers.5 Survivors’ increased risk for other chronic diseases (eg, type 2 diabetes) are exacerbated by unhealthy lifestyle behaviors.6,7 Likewise, survivors may experience both short- and long-term mental health problems, including adjustment disorders, anxiety, and depression.8-10 Survivorship care occurs after active treatment and includes the prevention and management of long-term and late effects of cancer and its treatment.11 The growing number of survivors makes attention to posttreatment disease prevention and management efforts paramount.
Follow-up care and surveillance are important elements of survivorship care research.12,13 The National Academy of Medicine and the American Society of Clinical Oncology have endorsed survivorship care plans (SCPs) as part of comprehensive quality care. SCPs are individualized plans that include a treatment summary, psychosocial support resources, lifestyle guidelines, a surveillance plan, and information regarding managing treatment-related side effects.12,14-17 SCPs are intended to integrate evidence-based guidelines and improve patient- centered care and coordination.12,15,18-20 However, both prior research and a recent systematic review indicate that the benefits of SCPs on several patient-reported outcomes are unclear or limited.21-25
While prior work reflects a necessary step in SCP research, significant limitations have been noted (eg, small samples, few randomized clinical trials, and heterogeneous methodology), and some evidence suggests that SCPs may be ineffective for improving patient-reported outcomes.25 Additionally, studies have failed to capture outcomes among patients at higher risk based on a family history of cancer. Work to date has not examined how SCPs are related to higher-risk survivors’ self-efficacy for disease coping, which is critical to disease and symptom management in cancer and other chronic disease populations. In addition, few studies have evaluated outcomes such as adherence to surveillance and lifestyle recommendations and appropriate uptake of these services in survivors at higher risk due to a family history.
The current study evaluated receipt of SCPs and associations between SCPs and psychosocial, quality of life, and health behavior outcomes among a sample of cancer survivors enriched with survivors with a family history of cancer. Our primary goal was hypothesis generation regarding the prevalence of SCP receipt and potential effects of survivorship care planning and follow-up in a higher-risk group. We hypothesized survivors who received SCPs would have better outcomes (eg, lower distress, higher self-efficacy/activation, and healthier lifestyles) compared with those who did not receive SCPs. Our findings have implications for the design of survivorship care interventions among individuals with a personal and family history of cancer.
Materials and Methods
Design
We conducted a cross-sectional online survey of adult cancer survivors who were diagnosed with cancer in the past 5 years and had completed active treatment.
Participants
Eligibility criteria included a cancer diagnosis (including recurrence) within the past 5 years; age ≥21 years; and English speaking. Exclusion criteria included undergoing treatment or inability to provide informed consent. Participants could report a range of risk for future cancer based on a self-reported family history of cancer. We define high risk based on participant-reported family history of having 2 or more family members with a diagnosis of cancer and/or personal history of a genetic mutation in self or other family members.
Recruitment
Participants were recruited from multiple sources, including Georgetown Lombardi Comprehensive Cancer Center Nontherapeutic Shared Resource (formerly the Familial Cancer Registry); advocacy/support organizations (eg, Colon Cancer Alliance, Susan G. Komen); Facebook; and newspaper advertisement. The range of recruitment sources allowed us to recruit a sample that included an overrepresentation of individuals at high risk based on self-reported family history of cancer. This oversampling was primarily due to our recruitment efforts through a registry at Georgetown Lombardi that included patients from families with hereditary cancer syndromes.
Procedure
We obtained institutional review board approval through Virginia Polytechnic Institute and State University. Online survey software approved for research was used to administer the informed consent and questionnaire. Following consent, participants completed the 30-minute survey. Participants had the option to enter a drawing for one of many $10 e-gift cards.
Measures
Sociodemographics
Participants reported their age, age at diagnosis, race, marital status, education, and household income.
Clinical Information
Participants provided data on personal cancer history (stage and treatment) and family cancer history.
SCPs
Participants were provided with definitions of an SCP and treatment summary and were asked whether they received an SCP, treatment summary, both, or neither. We defined an SCP as:
“A survivorship care plan is often considered a ‘wellness plan’ and is a comprehensive document that is used after treatment has been completed. Survivorship care plans include recommendations and referrals that are designed to improve a patient’s health and well-being, both physically and mentally. These plans also provide guidelines to monitor recovery as well as screen for recurrent and/or second cancers.”
We defined a treatment summary as:
“A treatment summary is typically a brief summary of cancer treatment. It is commonly used to organize information about a patient’s cancer, treatment, follow-up care, and basic information related to medical history and future doctors.”
Patient Satisfaction
Participants completed the Patient Satisfaction with Cancer-Related Care measure,26 which is 35 items assessing survivors’ satisfaction with cancer-related care. Item responses were summed to create a total score; lower scores indicate higher satisfaction (Cronbach’s α = .97).26
Quality of Life
The 27-item Functional Assessment of Cancer Therapy-General (FACT-G V4.0) assesses quality-of-life concerns specific to cancer patients and survivors.27 The FACT-G comprises 4 domains: (1) physical well-being; (2) social/family well-being; (3) emotional well-being; and (4) functional well-being.27 Higher scores indicate better quality of life (α = .93).27
Psychological and Cancer-Related Distress
The Brief Symptom Inventory (BSI) and Impact of Event Scale28 were used to measure psychological distress (α = .94 and .92, respectively). To reduce burden, the BSI-18 was condensed to 11 items.29
Coping Self-Efficacy
The 12-item Cancer Behavior Inventory-Brief30 assesses self-efficacy for coping with a cancer diagnosis in the following areas: maintaining independence and a positive attitude, actively participating in medical care, coping/stress management, and managing affect.30 Item responses were summed to create a total score for self-efficacy for coping with cancer-related tasks (α = .93).30
Patient Activation
The 13-item Patient Activation Measure31 assesses patient activation, defined by 4 stages: (1) believing the patient role is important; (2) having the confidence and knowledge to take action; (3) taking action to maintain and improve one’s health; and (4) staying the course even under stress. Raw scores were transformed to a scaled 0-100, with higher scores indicating greater self-management (α = .86).31
Health Behaviors
The Block and colleagues’32 brief screeners assessed postdiagnosis servings of dietary fats and fruits/vegetables. Item responses were summed to create a total fat score and total fruit/vegetable score (α = .80 and α = .58, respectively). Physical activity was assessed using the Stanford Leisure-Time Activity Categorical Item,33 which asks individuals to identify their level of engagement in physical activity during their leisure time in the past month.
Depression Screener
The Patient Health Questionnaire-234 is a 2-item yes/no screener for the frequency of depressive symptoms during the past 2 weeks: (1) feeling sad, blue, depressed, or down; and (2) lack of enjoyment/pleasure (α = .78). Responding “yes” to one or both was considered a positive screen.
Statistical Analyses
We computed descriptive analyses for sociodemographic variables and clinical health data. We examined the rate of participants receiving a written care plan (SCP or treatment summary). Independent Student’s t tests were used to evaluate whether demographic differences existed for those who received a written care plan versus those who did not. We used a 1-way between-groups multivariate analysis of covariance (MANCOVA) to investigate group differences in psychosocial and health behavior variables based on receipt of a written care plan. An empirical selection of covariates was used. Scores on the depression screen were highly correlated with many of the psychosocial and health behavior variables; thus, depression screen scores were included as a covariate in the MANCOVA.
Results
Sample Characteristics
Sociodemographic data are presented in Table 1. The final sample included 123 participants (109 women and 14 men; 93% Caucasian) from 34 states aged 23 to 74 years. The majority of the participants were married, had achieved at least a college degree, were employed full-time, and had an annual household income ≥$75,001.
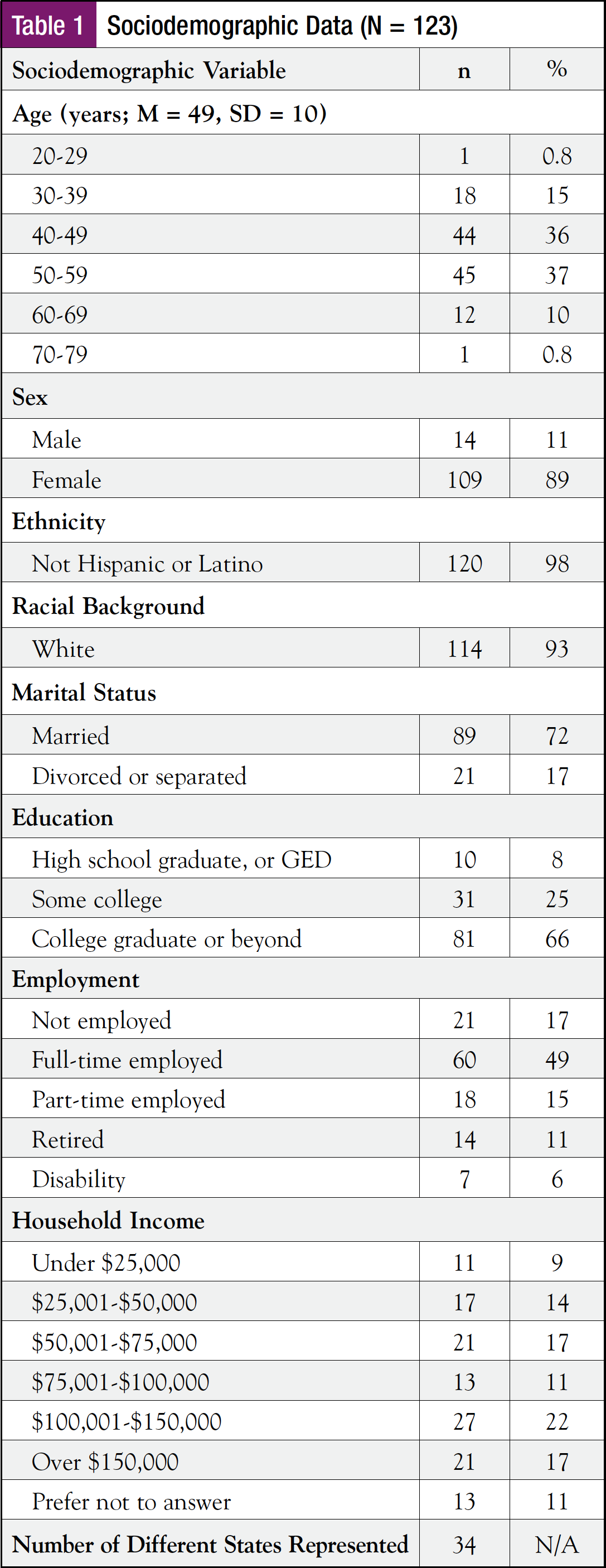
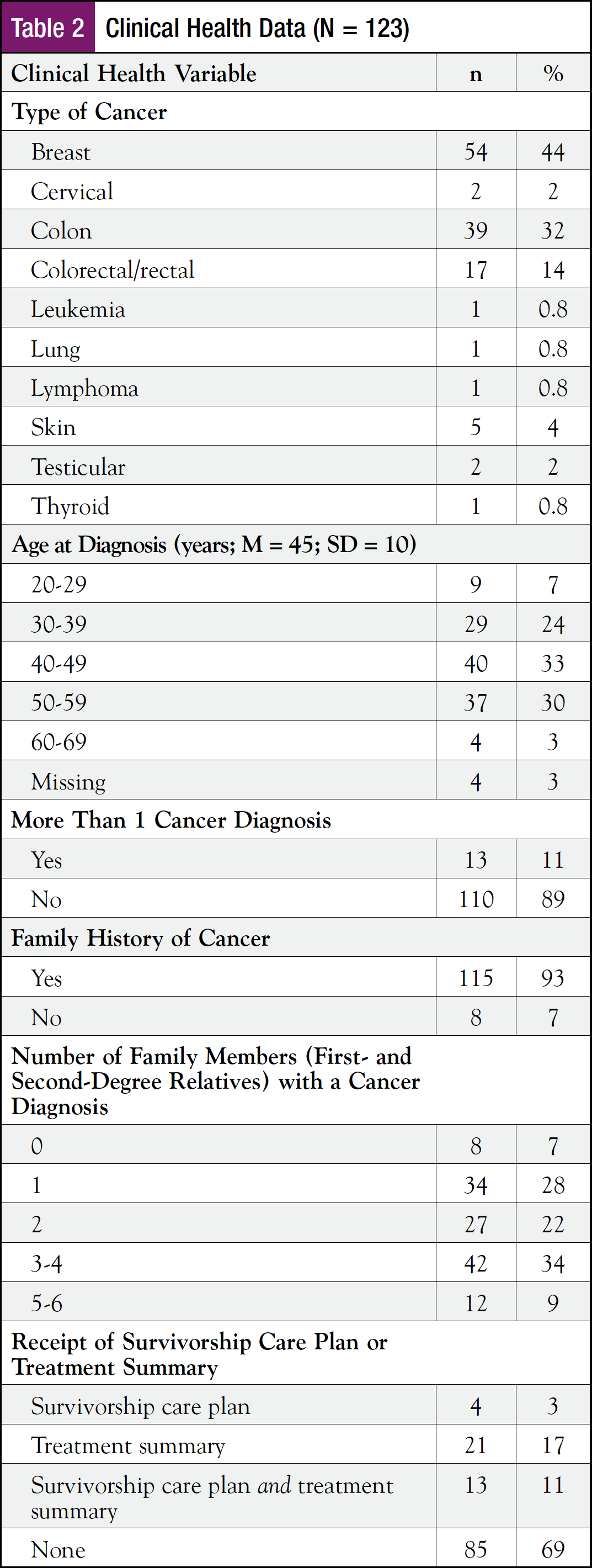
Clinical health data are presented in Table 2. The average age at diagnosis was 45 years (SD = 10), and 13 participants (11%) reported being diagnosed with >1 type of cancer. Overall, participants had been diagnosed with 10 different types of cancer. The most common cancer was breast (44%; n = 54), followed by colon (32%; n = 39). The majority of participants (93%; n = 115) reported a family history of cancer (M = 2 first- or second-degree relatives, range = 6).
Primary Outcomes
The majority of participants (69%; n = 85) reported not having received a written care plan (SCP or treatment summary). Among those who did (31%; n = 38), 3% (n = 4) received an SCP, 17% (n = 21) received a treatment summary, and 11% (n = 13) received both.
t Tests
The t tests were used to determine whether those who received a written care plan (n = 38) differed from those who did not. Due to the low number of patients receiving written care plans, we collapsed across groups. No significant differences were found for receipt of a written care plan across the demographic variables. Given the small number of men in the current sample, we used independent Student’s t tests to evaluate whether group differences existed between males and females on outcome variables. The only significant difference was on the Block Dietary Fat Screener t (117) = 2.56, P = .012, with men reporting greater fat intake (M = 21.38, SD = 6.08) than women (M = 5.73, SD = 7.67).
Multivariate Analysis of Covariance
We conducted a MANCOVA to investigate group differences in psychosocial and health behavior variables based on receipt of a written care plan. Nine dependent variables were used: patient satisfaction with cancer care, quality of life, general and cancer-specific distress, coping self-efficacy, patient activation, dietary fat intake, fruit/vegetable consumption, and physical activity. The independent variable was written care plan receipt, which included receipt of an SCP and/or treatment summary. After adjusting for depression scores (F [9, 104] = 9.857, P = .000), there was a significant overall effect of written care plan receipt on the dependent variables (F [9, 104] = 2.260, P = .024).
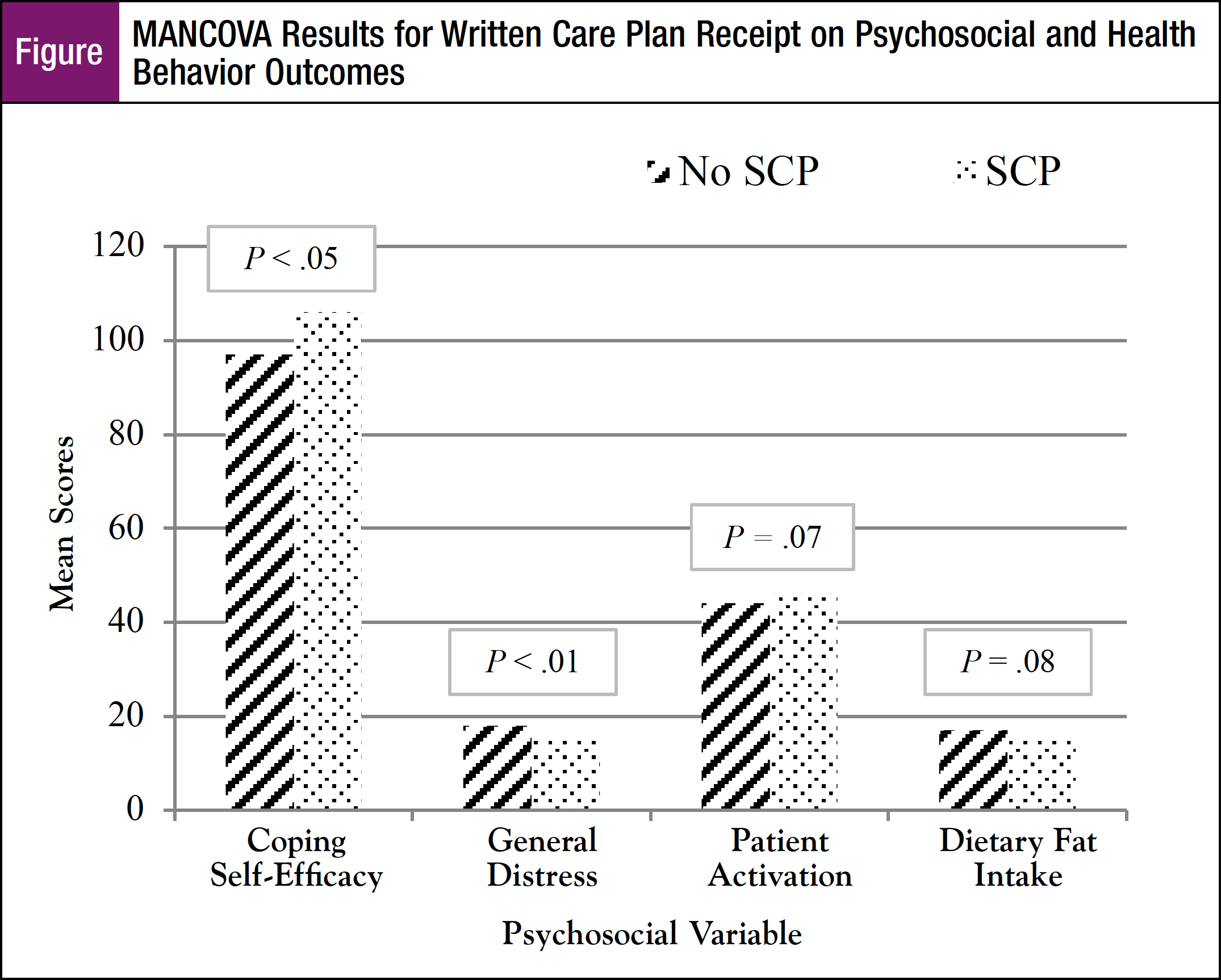
When the results for the psychosocial and health behavior dependent variables were considered separately, after controlling for depression, there were significant differences for general psychological distress (F [1, 104] = 8.316, P = .005) and coping self-efficacy (F [1, 104] = 6.627, P = .011). Depression-adjusted mean scores suggested that those who received a written care plan reported lower levels of general psychological distress (M = 14.96 vs M = 18.20) and higher levels of coping self-efficacy (M = 106.07 vs M = 96.49) compared with those who did not (Table 3, Figure). Finally, although not statistically significant, 2 potential trends were found for patient activation and dietary fat intake (P <.10 for each). Depression-adjusted mean scores indicated that those who received a written care plan were more likely to report greater levels of patient activation (M = 45.81 vs M = 43.79) and less dietary fat intake (M = 14.54 vs M = 17.26), compared with those who did not receive a written care plan.

Discussion
The current study explored the prevalence of SCP receipt and associations between receipt of a written care plan (SCP and/or treatment summary) and psychosocial and health behavior outcomes. To our knowledge, this study is among the first to evaluate written care plan receipt among a sample enriched with survivors at high risk based on family history and young age at diagnosis. Only a minority of participants reported receiving some form of written care plan. This finding speaks to the current state of survivorship care and SCPs—SCPs are rare and not uniformly implemented even among those at high risk based on personal and family history. Consistent with recent evidence, receipt of a written care plan was associated with significantly lower psychological distress and greater coping self-efficacy.25 Receipt of a written care plan was unrelated to patient satisfaction, quality of life, cancer-related distress, fruit/vegetable consumption, and physical activity. Patient activation and dietary fat intake showed slight differences between those who did and did not receive written care plans, although these differences were not statistically significant. Future work can explore whether differences emerge in these health outcomes with a larger sample of high-risk survivors.
Participants’ medical characteristics (ie, age at diagnosis and family history of disease) indicate our sample was high-risk, an important distinction from samples described in recent reviews.21-24 Specifically, individuals with an early age of cancer onset and/or a family cancer history tend to be at increased risk for recurrent and/or second primary cancers as well as long-term and late effects.35,36 Notably, the average age of cancer onset in the current sample (45 years) is significantly younger than people at average risk (65 years).37 Young adult cancer survivors have been shown to have poor adherence to follow-up care,38 which may stem from many factors, including a lack of understanding of care recommendations,38-40 being uninsured or underinsured,41,42 age-specific competing demands (eg, school, work, childcare),38,43,44 and being a highly mobile population. Coupled with the high rate of participants with a family history of cancer (93%), this highlights the at-risk nature of the current group. As such, study participants were potentially in need of a higher level of survivorship care to help them effectively manage and reduce their risk of recurrent and/or second primary tumors, mitigate and address long-term and late effects of cancer treatments, and address psychosocial issues related to cancer.
In a randomized clinical trial to determine if an SCP for breast cancer survivors improves patient-reported outcomes, no differences were found in the outcomes under investigation (eg, general psychological distress, quality of life, patient satisfaction) when comparing SCPs with standard discharge/transfer visits at follow-up.45 Two recent reviews similarly found little impact of SCPs on a variety of patient-reported and health outcomes.21,25 Consistent with results from the systematic review by Hill and colleagues,25 results from the current study reveal significantly lower general distress and higher coping self-efficacy for those who received a written care plan. Differences between the prior and current studies include possible lack of measurement sensitivity (ie, outcome measures other than the Impact Event Scale may be more sensitive to the effects of SCPs), the breast cancer–specific survivor sample (eg, breast cancer patients may be an especially well-informed survivor group), and the fact that some of the research was conducted in Canada where healthcare is universal and standard of care differs.45
Despite growing momentum to improve survivorship care and care planning, cancer centers and survivorship interventions continue to encounter obstacles related to the delivery of quality care.46 Our results also raise questions about implementation and adherence to recommended and established guidelines related to survivorship care. For example, recent guidelines suggest provision of survivorship care, rather than a single written document (Commission on Cancer [CoC], Standard 4.9).47,48 Previously, a study of survivorship centers found that SCPs included less than two-thirds of content recommendations,46 with very few content domains (8/23) consistently addressed by centers.46 Among the content areas most often omitted were familial cancer risk information, signs of recurrent/secondary cancers, supportive care, clinical trials, and contact information for a care coordinator.46 The challenges of addressing the recommended content areas and SCP implementation potentially outweigh the perceived benefits.49 Some of the most notable challenges include personnel, time and resource constraints, and fragmentation in survivorship care. Current efforts to identify survivorship care as a process, with attention to survivors’ needs over time, are needed.47 A useful next step in this line of research is to examine the use of SCPs as a “tool” or “springboard” for ongoing conversations with and monitoring by the patient’s medical team, particularly their survivorship nurse and/or patient navigator. Such an approach aligns with the CoC recommendation to focus on survivorship care programs rather than SCPs alone.47,48
Our results also demonstrated that the difference in distress scores for those who received a written care plan versus those who did not was more than half a standard deviation. If replicated in future prospective research, these differences could be considered clinically meaningful.50 Future work can further explore the trends we identified that suggested receipt of a written care plan might be associated with higher patient activation and lower dietary fat intake. These findings provide initial support for written care plans for lower distress and higher coping self-efficacy in a high-risk sample of survivors. Written care plans may provide a potential starting point to optimize survivorship care in ways that promote disease management and risk reduction.51,52 For example, psychological distress in cancer survivors is associated with nonadherence to treatment recommendations, poorer satisfaction with care, and poorer quality of life across many domains,53 even though we have evidence-based strategies for identifying and mitigating distress.54,55
Likewise, survivors’ distress and coping self-efficacy have implications for communication with providers, disease and symptom management, psychosocial adjustment, seeking/understanding medical information, and adherence to follow-up care.56-58 The current findings suggest that SCPs may play a role in decreasing distress and improving self-efficacy. However, well-powered comparative effectiveness studies are needed to determine the true impact of SCPs on these important outcomes and whether these effects are sustained over time.25 Written care plans may therefore be an additional strategy for targeting psychosocial health and potentially have subsequent impacts on adherence with care and disease management, quality of life, and risk reduction during survivorship. Future research efforts should consider inclusion of outcomes that more closely align with the goals and processes of delivering SCPs (eg, self-efficacy, patient activation). Oncology/survivorship nurses and patient navigators are likely well positioned to engage patients in discussion of SCPs, and future research can explore the benefits of targeting efforts toward survivors at higher risk, including younger survivors and those with a familial cancer history. Additionally, next steps in research should leverage technology for SCP implementation and delivery for a low-cost and low-burden approach that includes patient self-reporting and tracking (eg, symptom monitoring, use of Bluetooth devices, integration with electronic health records to provide real-time data to providers), and involves opportunities for nurses and patient navigators to monitor, provide feedback, or initiate clinical action.59-61 This type of technology-based symptom self-reporting and monitoring is associated with notable clinical benefits during cancer care.59-61 Nurses and patient navigators can play an integral role in this process because they serve as a liaison between patients and their medical teams and can facilitate referrals for survivorship services based on patients’ self-report. Building a more consistent and rigorous evidence base in these areas will improve our understanding of (1) the most appropriate use for SCPs (eg, risk-based survivorship care62); (2) patient outcomes to target (eg, psychological distress, self-efficacy); and (3) delivery strategies that reduce provider burden and increase patient access.
This study has several strengths, including assessment of an important phase of cancer care that involves empirical evaluation of SCPs. Additionally, data were representative of people within various cancer survivor disease groups, age ranges, and geographical locations. Limitations should be noted. Based on sample size and homogeneous demographic characteristics, results may not be generalizable to male cancer survivors, survivors who are nonwhite or those with lower socioeconomic status. Participants were primarily breast and colorectal cancer survivors, so results also may not generalize to patients with other disease types. Participants may have been more highly motivated and more compliant with lifestyle behavioral recommendations given their potentially higher risk status based on young age at diagnosis and family history. Finally, the cross-sectional design prohibits causal inference; future work should assess these constructs over time.
Attention to survivorship care is a critical issue for survivors’ short- and long-term health and well-being.12,15,18 Further research is needed to examine the effectiveness of SCPs so that researchers and clinicians can better understand the effects of survivorship care strategies on survivor outcomes. Advancements in our knowledge and understanding of the needs of cancer survivors, survivorship care approaches, and whether SCPs are effective will allow healthcare professionals to provide high-quality evidence-based care.18 This study contributes to the evolving field of research on SCPs by focusing on a high-risk sample of cancer survivors. Future research should examine the overall effectiveness and long-term impact of SCPs among high-risk survivors on patient behaviors, adherence to surveillance recommendations, and clinical outcomes.
Acknowledgments: This study was funded by The Graduate Student Assembly at Virginia Polytechnic Institute and State University. This work was supported in part through shared resources and the Survivorship Research Initiative at Georgetown Lombardi Comprehensive Cancer Center (P30CA051008; Weiner, PI). The work of Joseph G. Winger was supported by a Kornfeld Scholars Program Award from the National Palliative Care Research Center.
We thank the Graduate Student Assembly at Virginia Polytechnic Institute and State University for funding this study as part of the Graduate Research Development Program. We wish to acknowledge the valuable contributions of Brenda M. Davy, PhD; Jack W. Finney, PhD; David W. Harrison, PhD; and Sarah Lamb, BS.
Ethical Standards: We obtained internal review board approval through Virginia Polytechnic Institute and State University. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the 1964 Declaration of Helsinki (5). Informed consent was obtained from all patients for being included in the study.
Conflict of Interest: The authors declare that they have no conflict of interest.
Disclosures: None.
Rights to Data: The authors have full control of all primary data and agree to allow the journal to review the data if requested.
References
- Bluethmann SM, Mariotto AB, Rowland JH. Anticipating the “Silver Tsunami”: prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiol Biomarkers Prev. 2016;25:1029-1036.
- Miller KD, Siegel RL, Lin CC, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66:271-289.
- American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2016-2017. Atlanta, GA: American Cancer Society; 2016.
- Miller KD, Nogueira L, Mariotto AB, et al. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin. 2019;69:363-385.
- Stein KD, Syrjala KL, Andrykowski MA. Physical and psychological long-term and late effects of cancer. Cancer. 2008;112(11 Suppl):2577-2592.
- Irwin ML, Mayne ST. Impact of nutrition and exercise on cancer survival. Cancer J. 2008;14:435-441.
- Winzer BM, Whiteman DC, Reeves MM, Paratz JD. Physical activity and cancer prevention: a systematic review of clinical trials. Cancer Causes Control. 2011;22:811-826.
- Derogatis LR, Morrow GR, Fetting J, et al. The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249:751-757.
- Miovic M, Block S. Psychiatric disorders in advanced cancer. Cancer. 2007;110:1665-1676.
- Stark D, Kiely M, Smith A, et al. Anxiety disorders in cancer patients: their nature, associations, and relation to quality of life. J Clin Oncol. 2002;20:3137-3148.
- Denlinger CS, Carlson RW, Are M, et al. Survivorship: introduction and definition. Clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2014;12:34-45.
- Hewitt M, Greenfield S, Stovall E, eds. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington, DC: National Academies Press; 2006.
- Levit LA, Balogh EP, Nass SJ, Ganz PA, eds. Delivering High-Quality Cancer Care: Charting a New Course for a System in Crisis. Washington, DC: The National Academies Press; 2013.
- American Society of Clinical Oncology. ASCO Cancer Treatment and Survivorship Care Plans. www.cancer.net/survivorship/follow-care-after-cancer-treatment/asco-cancer-treatment-and-survivorship-care-plans. Accessed November 16, 2020.
- Ganz PA, Casillas J, Hahn EE. Ensuring quality care for cancer survivors: implementing the survivorship care plan. Semin Oncol Nurs. 2008;24: 208-217.
- Jacobs LA, Palmer SC, Schwartz LA, et al. Adult cancer survivorship: evolution, research, and planning care. CA Cancer J Clin. 2009;59:391-410.
- National Cancer Institute. Follow-up medical care. www.cancer.gov/about-cancer/coping/survivorship/follow-up-care. Updated November 4, 2020. Accessed November 16, 2020.
- Earle CC. Failing to plan is planning to fail: improving the quality of care with survivorship care plans. J Clin Oncol. 2006;24:5112-5116.
- Faul LA, Shibata D, Townsend I, Jacobsen PB. Improving survivorship care for patients with colorectal cancer. Cancer Control. 2010;17:35-43.
- Hahn EE, Ganz PA. Survivorship programs and care plans in practice: variations on a theme. J Oncol Pract. 2011;7:70-75.
- Brennan ME, Gormally JF, Butow P, et al. Survivorship care plans in cancer: a systematic review of care plan outcomes. Br J Cancer. 2014;111: 1899-1908.
- Mayer DK, Birken SA, Check DK, Chen RC. Summing it up: an integrative review of studies of cancer survivorship care plans (2006-2013). Cancer. 2015;121:978-996.
- van de Poll-Franse LV, Nicolaije KAH, Ezendam NPM. The impact of cancer survivorship care plans on patient and health care provider outcomes: a current perspective. Acta Oncol. 2017;56:134-138.
- LaGrandeur W, Armin J, Howe CL, Ali-Akbarian L. Survivorship care plan outcomes for primary care physicians, cancer survivors, and systems: a scoping review. J Cancer Surviv. 2018;12:334-347.
- Hill RE, Wakefield CE, Cohn RJ, et al. Survivorship care plans in cancer: a meta-analysis and systematic review of care plan outcomes. Oncologist. 2020;25:e351-e372.
- Jean-Pierre P, Fiscella K, Freund KM, et al. Structural and reliability analysis of a patient satisfaction with cancer-related care measure: a multisite patient navigation research program study. Cancer. 2011;117:854-861.
- Cella DF, Tulsky DS, Gray G, et al. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol. 1993;11:570-579.
- Horowitz M, Wilner N, Alvarez W. Impact of Event Scale: a measure of subjective stress. Psychosom Med. 1979;41:209-218.
- Peshkin BN, Demarco TA, Graves KD, et al. Telephone genetic counseling for high-risk women undergoing BRCA1 and BRCA2 testing: rationale and development of a randomized controlled trial. Gen Test. 2008;12:37-52.
- Heitzmann CA, Merluzzi TV, Jean-Pierre P, et al. Assessing self-efficacy for coping with cancer: development and psychometric analysis of the brief version of the Cancer Behavior Inventory (CBI-B). Psychooncology. 2011;20:302-312.
- Hibbard JH, Mahoney ER, Stockard J, Tusler M. Development and testing of a short form of the patient activation measure. Health Serv Res. 2005;40(6 Pt 1):1918-1930.
- Block G, Gillespie C, Rosenbaum EH, Jenson C. A rapid food screener to assess fat and fruit and vegetable intake. Am J Prev Med. 2000;18:284-288.
- Kiernan M, Schoffman DE, Lee K, et al. The Stanford Leisure-Time Activity Categorical Item (L-Cat): a single categorical item sensitive to physical activity changes in overweight/obese women. Int J Obesity (Lond). 2013;37:1597-1602.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Baglia ML, Tang MTC, Malone KE, et al. Family history and risk of second primary breast cancer after in situ breast carcinoma. Cancer Epidemiol Biomarkers Prev. 2018;27:315-320.
- Hemminki K, Li X, Dong C. Second primary cancers after sporadic and familial colorectal cancer. Cancer Epidemiol Biomarkers Prev. 2001;10:793-798.
- The National Cancer Institute. Age and cancer risk. www.cancer.gov/about-cancer/causes-prevention/risk/age. Published April 29, 2015. Accessed November 16, 2020.
- Smits-Seemann RR, Kaul S, Zamora ER, et al. Barriers to follow-up care among survivors of adolescent and young adult cancer. J Cancer Surviv. 2017;11:126-132.
- Keegan THM, Lichtensztajn DY, Kato I, et al. Unmet adolescent and young adult cancer survivors information and service needs: a population-based cancer registry study. J Cancer Surviv. 2012;6:239-250.
- Casillas J, Syrjala KL, Ganz PA, et al. How confident are young adult cancer survivors in managing their survivorship care? A report from the LIVESTRONG Survivorship Center of Excellence Network. J Cancer Surviv. 2011;5:371-381.
- Keegan THM, Tao L, DeRouen MC, et al. Medical care in adolescents and young adult cancer survivors: what are the biggest access-related barriers? J Cancer Surviv. 2014;8:282-292.
- Parsons HM, Schmidt S, Harlan LC, et al. Young and uninsured: insurance patterns of recently diagnosed adolescent and young adult cancer survivors in the AYA HOPE study. Cancer. 2014;120:2352-2360.
- Miedema B, Easley J. Barriers to rehabilitative care for young breast cancer survivors: a qualitative understanding. Support Care Cancer: 2012; 20:1193-1201.
- Rabin C, Simpson N, Morrow K, Pinto B. Intervention format and delivery preferences among young adult cancer survivors. Int J Behav Med. 2013;20:304-310.
- Grunfeld E, Julian JA, Pond G, et al. Evaluating survivorship care plans: results of a randomized, clinical trial of patients with breast cancer. J Clin Oncol. 2011;29:4755-4762.
- Stricker CT, Jacobs LA, Risendal B, et al. Survivorship care planning after the Institute of Medicine recommendations: how are we faring? J Cancer Surviv. 2011;5:358-370.
- American College of Surgeons. Commission on Cancer. DRAFT Revised Standards. www.facs.org/~/media/files/quality%20programs/cancer/coc/draft_coc_revised_standards_may2019.ashx. Published 2019. Accessed June 16, 2020.
- American College of Surgeons. Commission on Cancer. Crosswalk Between 2016 and 2020 CoC Standards. www.facs.org/-/media/files/quality-programs/cancer/coc/crosswalk_between_2016_and_2020_coc_standards.ashx. Published 2020. Accessed June 16, 2020.
- Salz T, Oeffinger KC, McCabe MS, et al. Survivorship care plans in research and practice. CA Cancer J Clin. 2012;62:101-117.
- Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41:582-592.
- Nekhlyudov L, Galioto M. Innovations in cancer survivorship care: “Lessons from the Clinic” special section. J Cancer Surviv. 2020;14:1-3.
- Goldenberg BA, Carpenter-Kellett T, Gingerich JR, et al. Moving forward after cancer: successful implementation of a colorectal cancer patient- centered transitions program. J Cancer Surviv. 2020;14:4-8.
- Jacobsen PB, Donovan KA, Trask PC, et al. Screening for psychologic distress in ambulatory cancer patients. Cancer. 2005;103:1494-1502.
- Hahn EE, Munoz-Plaza CE, Pounds D, et al. Pragmatic randomized trial of a distress screening program using an effectiveness-implementation hybrid design. J Clin Oncol. 2019;37(suppl 5). Abstract 5.
- Ehlers SL, Davis K, Bluethmann SM, et al. Screening for psychosocial distress among patients with cancer: implications for clinical practice, healthcare policy, and dissemination to enhance cancer survivorship. Transl Behav Med. 2019;9:282-291.
- Mosher CE, Duhamel KN, Egert J, Smith MY. Self-efficacy for coping with cancer in a multiethnic sample of breast cancer patients: associations with barriers to pain management and distress. Clin J Pain. 2010; 26:227-234.
- Philip EJ, Merluzzi TV, Zhang Z, Heitzmann CA. Depression and cancer survivorship: importance of coping self-efficacy in post-treatment survivors. Psychooncology. 2013;22:987-994.
- Shields CG, Ziner KW, Bourff SA, et al. An intervention to improve communication between breast cancer survivors and their physicians. J Psychosoc Oncol. 2010;28:610-629.
- Basch E, Charlot M, Dueck AC. Population level evidence of survival benefits of patient reported outcome symptom monitoring software systems in routine cancer care [published online October 7, 2020]. Cancer Med.
- Basch E, Deal AM, Dueck AC, et al. Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA. 2017;318:197-198.
- Basch E, Deal AM, Kris MG, et al. Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol. 2016;34:557-565.
- Kline RM, Arora NK, Bradley CJ, et al. Long-term survivorship care after cancer treatment – summary of a 2017 National Cancer Policy Forum Workshop. J Natl Cancer Inst. 2018;110:1300-1310.



