Abstract: Cognitive and behavioral therapy techniques have been found to be helpful in treating insomnia and other sleep disorders. Higher rates of insomnia occur in the cancer population than the general public. This case reviews the conventional techniques used to treat insomnia, as well as the alterations made to treat a patient due to characteristics and the less than the traditional time frame of the treatment.
This case study is a breast cancer survivor who was referred for treatment for her anxiety and insomnia. The patient had several unique factors that needed to be considered during assessment and treatment planning: cancer history, single parent, ethnicity, temperament, education level, gender, age, and work status. Additional barriers to care that are addressed include the patient’s lack of desire to stop certain sleep habits, her lack of interest in keeping sleep logs, and place as well as the frequency of the clinic encounters. With only 4 sessions of cognitive behavioral therapy, the patient reported decreased anxiety and insomnia.
Introduction
Insomnia is characterized by dissatisfaction with the quality or quantity of sleep, difficulty maintaining or initiating sleep, or waking too soon and being unable to reinitiate sleep.1 An estimated one-third of adults describe experiencing symptoms of insomnia, with 6% to 15% meeting the criteria for insomnia.1,2 Many factors may predispose individuals to develop sleep disturbances due to alterations in the normal sleep regulatory process, such as lifestyle factors, psychological components, demographics, and experiencing a major life event.1,3-8 Chronic insomnia has been shown to have negative impacts on the individual and the larger society due to higher healthcare utilization, increased difficulty with job performance and attendance, and reduced quality of life.9,10
The role of cognitive and behavioral therapy interventions for the treatment of sleep disturbances has been well studied and documented.11 Meta-analyses continue to support the effectiveness of these techniques in treating insomnia, specifically with regard to improving sleep latency, sleep quality, sleep efficiency, total sleep time, and awakenings after sleep onset.12-18
Specific cognitive and behavioral therapy techniques often used in treating insomnia include relaxation, sleep hygiene, sleep scheduling, paradoxical intention, and cognitive techniques. Relaxation techniques are used based on the idea that the reduction of tension and the anxiety that may be related to it will help promote sleep. Sleep hygiene19 includes lifestyle behaviors focused on improving sleep, often presented as rules or tips such as limiting caffeine before bed and not napping during the day. Sleep scheduling utilizes sleep restriction and stimulus control to reassociate the bed and bedtime with sleep rather than being awake. Paradoxical intention instructs the patient to exaggerate the symptoms they have a problem with instead of trying to change it; with insomnia, this includes purposefully trying to stay awake at night.20 Cognitive techniques focus on identifying, challenging, and changing dysfunctional attitudes and beliefs that create or maintain sleep difficulties.
Because experiencing a major life event has been identified as a predisposition to insomnia,1,8 it is not surprising that cancer patients have higher rates of insomnia than the general population.21-24 Cognitive behavioral therapy (CBT) has repeatedly been shown to effectively treat insomnia in cancer patients.25
The present report describes a case study of Mary (pseudonym), a female in her early 50s who was born and raised in China but moved to the United States in her late teens. This article will elaborate on the hypothesis that CBT was an appropriately matched treatment for Mary. In addition, this article will describe the course and outcome of treatment, the real-life challenges that arise when attempting to utilize a manualized treatment, as well as specifically address the unique factors that contributed to this case.
Case Description
Mary was diagnosed with breast cancer by her gynecologist when she was in her late 40s. Her full cancer diagnosis was a 2.7-cm, grade 3, lymphovascular invasion–positive, hormone receptor–positive, HER2-negative cancer with 1:2 positive lymph nodes, stage IIB breast cancer. A month after her cancer diagnosis she had a left breast total skin-sparing mastectomy with immediate reconstruction with 300-mL tissue expander, serratus muscle rotation, and sentinel lymph node mapping and biopsy. A month after her surgery, she began 6 rounds of chemotherapy that lasted a total of 5 months. Following chemotherapy, she started taking an aromatase inhibitor, which she would need to take for 5 to 10 years total. Seven months after her initial diagnosis, Mary began 6 weeks of radiation therapy. Eighteen months after her diagnosis, she had her left expander exchanged for a saline implant.
Mary was previously married; her husband died of cancer 4 years ago. She is a retired dentist and has a teenage daughter who she is raising on her own. Her mother died when she was 10 years of age, and her father died of acute leukemia when she was age 23. She has 1 brother who is alive and healthy. Mary has low social support, but she is physically active—walks daily, even during her entire cancer treatment.
Mary reported experiencing 2 other episodes of anxiety/sleep problems in her past but never sought treatment, noting that they resolved on their own. She also reported experiencing some social anxiety, but again did not seek treatment. She denied any other significant mental health history.
Case Context and Method
Mary was referred to a cancer center psychology clinic by her oncologist for assistance with anxiety and sleep issues 2 years after her cancer diagnosis. The referral noted:
“Her primary concern today is that of insomnia and anxiety. She states that prior to her recent surgery she became extremely anxious and worried about complications related to the surgery. Since this time her anxiety level has not decreased. She has been taking lorazepam 0.25-0.5 mg at night and has been able to sleep. However, on the days when she does not take lorazepam, she is awake all night. She then has difficulty performing her daily activities, which focus on the care of her daughter.”
Mary describes a “vicious cycle about worrying I will not be able to go to sleep—no sleeping—feeling anxious.” She reported that her anxiety and sleep issues started the week before her breast implant replacement surgery when she would wake up 2 to 3 times per night with anxiety about the surgery. “I was afraid that this is the last leg of my journey and that something would go wrong.” She stated that her surgery went well but she continued to experience anxiety and insomnia. She spoke with her primary care provider (PCP) and explained she had 1.0-mg strength lorazepam from her chemotherapy (which she was prescribed and had filled at the time of chemotherapy but did not take). Through her PCP’s encouragement, she started taking 0.5 mg of lorazepam before bed for 4 to 5 days and was able to successfully sleep. However, Mary worried about taking any medications and thus stopped the lorazepam and again could not sleep and “felt bad...anxious all day about if I would sleep.” Mary experimented with a lower dose (0.25 mg) of lorazepam and found that dose to generally be helpful for her anxiety and sleep problems as well. At the time of initial assessment, she was vacillating between taking the lorazepam and sleeping versus trying to not take the lorazepam and subsequently not sleeping. Additionally, she noted that she spent her entire day worrying about whether to take the medication, whether she would sleep, and how she would function the next day if she did not sleep. Mary expressed fear that her lack of sleep and anxiety would make her cancer come back and stated that focusing on her sleep and anxiety is her “number 1 priority” at this time.
Case Formulation and Treatment Approach
Mary’s depressive and anxiety symptoms were assessed and measured with the Patient Health Questionnaire 9-item depression scale (PHQ-926) and the Generalized Anxiety Disorder 7-item scale (GAD-727), respectively. She scored 1 on the PHQ-9, indicating no elevated depressive symptoms, and scored 13 on the GAD-7, indicating moderate levels of anxiety.
Mary had a prescription for lorazepam but was interested in nonpharmacologic treatments for her anxiety and sleep. Other factors important to the case conceptualization included her cultural background, current age/the changing nature of sleep and sleep needs based on changing needs over the lifespan, history of cancer treatments (including multiple surgeries, chemotherapy, and radiation), current use of aromatase inhibitors to prevent cancer recurrence (which she would likely be on for several more years), current physical activity level, daytime schedule/lack of currently working, and beliefs that her lack of sleep and anxiety would cause her cancer to recur. Given the interrelational nature of Mary’s insomnia and anxiety and the individual considerations described above, it was determined that she would benefit from CBT for her insomnia, using both behavioral techniques to help recondition her body to sleep, as well as cognitive components are given her strong beliefs about sleep.
Course of Treatment and Monitoring of Progress
Session 1
Mary was skeptical that nonmedication interventions would help her sleep; however, she was willing to try this care. The initial sleep assessment was bypassed, and the sleep intervention was started in the first session. The session focused on providing Mary with psychoeducation related to sleep, a rationale for the cognitive and behavioral therapies to be used, sleep hygiene and stimulus control (including using the bed only for sleep/sex, not taking naps during the day), and information on sleep restriction. Estimating her time in bed for the past few nights, Mary’s estimated sleep efficiency was calculated:
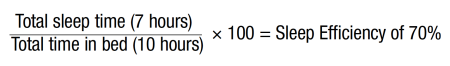
Using this sleep efficiency information, sleep restriction started by creating a set bedtime (11:00 pm) and set wake-up time (6:00 am) that matched Mary’s current total sleep time. She had concerns about not being able to sleep during the designated sleep time and therefore be nonfunctional the following day when she needed to be alert to drive her daughter to activities. However, she voiced understanding for the rationale and commitment to follow the set bedtime and wake-up times as well as the no-napping rule. To work with Mary’s cognitions related to sleep (she will not be able to sleep, and her lack of sleep will cause her cancer to return), the session included some basic cognitive work, including information that there is no consistent research to suggest that her sleep issues will cause a cancer recurrence. She was given permission not to sleep (paradoxical intention) and reminded of times when she did not sleep but was able to function the next day. Identifying maladaptive thinking and changing it to adaptive thinking through cognitive restructuring was also briefly discussed.
Session 2
Mary returned 3 weeks later. She reported that her anxiety was gone, and she was now sleeping regularly every night. She had employed the sleep restriction and part of the stimulus control discussed in the prior session: no naps during the day, only in bed from 11:00 pm until 6:00 am. Mary was engaging in non-sleep/sex activities in the bed but found that she slept well the first night, and her anxiety about sleep was gone because she swapped her maladaptive thinking (“I should be asleep” and “I won’t be able to get to sleep”) to adaptive thinking (“If I don’t sleep well tonight I know I will tomorrow”). Her estimated current sleep efficiency was calculated:
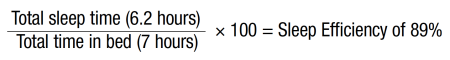
Given her sleep efficiency score of 89%, she was encouraged not to change her time in bed but continue with her total time in the bed of 7 hours (11:00 pm until 6:00 am) and then reassess at the next appointment. To better track her sleep, Mary was provided with a sleep log to fill out daily, but she reported that she was not willing to try it.
Given her success with sleep restriction, time was spent in the second session reassessing the areas that are still causing Mary difficulties with her sleep. She was able to identify additional challenges to her sleep, including attempting to “will” herself back to sleep, looking at the alarm clock multiple times when she cannot sleep, and having stressful thoughts while lying awake in bed. She was encouraged to practice diaphragmatic breathing (as practiced in session) during the day to gain mastery so she can eventually implement it at night to help redirect her focus when she is having stressful thoughts. She was encouraged to turn her alarm clock around at night so she is not able to look at it. Additional stimulus control techniques were discussed, including getting out of bed after 20 minutes of not sleeping and sitting in a chair to read a boring/neutral magazine, only getting back in bed when she is drowsy.
Session 3
Mary returned 4 weeks later. She reported that her anxiety continued to be absent, and she was reliably sleeping every night. She had altered her bedtime/wake-up time; she was now going to bed around 10:00 or 10:30 pm and getting out of bed around 6:30 or 7:00 am. Mary was not looking at her clock in the middle of the night, but instead reminding herself “it’s OK if I don’t sleep anymore tonight...I’ll be tired and sleep tomorrow night,” and trying diaphragmatic breathing for relaxation. She reported that the diaphragmatic breathing had not been helpful for her in getting back to sleep, but instead the cognitive component of reminding herself that it is “OK” if she does not sleep anymore was the most beneficial in helping her continue to stay anxiety free and fall back asleep. Regarding the stimulus control technique of getting out of bed after 20 minutes of not sleeping, Mary had not engaged in this technique because she felt it was “silly.” Her estimated current sleep efficiency was calculated using the following equation:
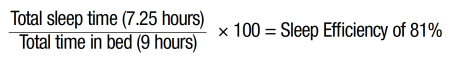
Given the sleep efficiency score of 81%, the idea of engaging in further sleep restriction to attempt to limit her awake time at night was discussed, but she was opposed to changing her bedtime and wake-up time.
Session 4
Mary returned 4 weeks later. She reported that her sleep continued to be much improved—only experienced difficulty falling asleep 2 nights during the prior 2 weeks. Her anxiety was also much improved. The following calculation estimated her current sleep efficiency:
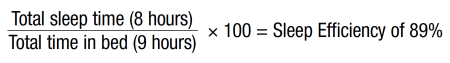
Mary’s estimated sleep efficiency was 89%, and she was happy with her current sleep schedule. She continued to avoid napping but was not engaging in the stimulus control technique of getting out of bed after 20 minutes of not being able to sleep. She noted that she was also continuing to read, watch TV, and use her tablet in bed, and she was not interested in changing those behaviors. She was continuing to try to practice diaphragmatic breathing when having difficulties returning to sleep but was having difficulty mastering the technique. Mary noted that she does “7 cleansing breaths” every morning to help her center herself, and therefore it was decided that she could use this type of breathing exercise for relaxation in bed. She was optimistic about the use of this relaxation technique to help her return to sleep at night when she has difficulties.
Treatment Outcome
Mary failed to return for a follow-up. When called to attempt to reschedule a canceled appointment, she stated that she was “doing well” and continuing to feel good about her sleep and her anxiety. She did not feel that further follow-up was necessary at this time.
Discussion
During the course of work with Mary, it was important to consider her unique factors. The below list of factors not only applies to Mary but should be considered when developing individual treatment plans.
Cancer History
The distress of a cancer diagnosis can precipitate insomnia.7 Many of Mary’s automatic thoughts were of her sleep and anxiety causing a cancer recurrence and ultimately leaving her daughter orphaned without either parent. Approximately 40% of breast cancer survivors believe that stress caused their cancer, and about 30% believe that their cancer did not come back because they reduced their stress; however, there is no known research linking stress and insomnia as causes of breast cancer or cancer recurrence.28 Receiving cancer treatments such as surgery, chemotherapy, and radiation therapy have been correlated with sleep issues in cancer patients.7 During her various cancer treatments (surgeries, chemotherapy, and radiation), Mary had been encouraged by her healthcare professionals to nap, lie down in bed to rest, and get a lot of sleep. While those sleep and energy conservation suggestions may have been helpful at the time, the suggestions may have contributed to inaccurate beliefs and attitudes about sleep needs and the sleep-wake cycle, as well as disassociated sleep cycle–inducing cues, further perpetuating insomnia. Now considered the standard of care for certain types of breast cancer, Mary was taking an aromatase inhibitor; half of breast cancer patients on aromatase inhibitors experience insomnia.29
Being a Single Parent
Central in Mary’s concerns about her sleep issues is her role as a single mother. Being a widower put her at higher risk for developing insomnia.3,4,6 Although she had experienced problems with sleep and anxiety in the past, those were during times when her husband was alive and she was not a single parent. Mary was afraid that she might be too tired to safely drive her daughter to school/activities, and the fear of putting her daughter’s health at risk increased her anxiety even more; it was likely that her anxiety about putting her daughter’s life at risk would automatically decrease as her sleep improved and her fear of not being able to drive her daughter to school/activities decreased. Given that she is an only parent, the sooner Mary could notice changes in her sleep, the sooner she could relieve this very critical anxiety about her daughter.
Ethnicity
As someone of Asian descent, research suggests that Mary was less likely to talk with her healthcare providers about her sleep issues, be diagnosed with a sleep disorder, and be asked about her sleep by a healthcare provider than someone who is white, black, or Hispanic.5 Additionally, people of Asian descent may be more likely to use nonpharmaceutical techniques and aids to help them with sleep issues than they are to use pharmaceutical sleep aids.5
Temperament
Mary’s original referral included anxiety; her GAD-7 score on the day of the initial session was 13, indicating moderate levels of anxiety. Research suggests that individuals with anxious temperaments are at a higher risk for developing insomnia.1
Educational Level
Mary was a retired dentist, and her educational level helped determine the intensity of information she could be provided in session.
Gender
Being a woman, Mary was at an increased risk for insomnia and sleep-related complaints.1,3,4,6 Not surprisingly, sleep is affected by hormonal changes. Over her life span, Mary had many different hormonal changes that contribute to increased difficulty sleeping: puberty, menstruation, being pregnant with her daughter, and her cancer-induced menopause.30
Age
Research suggests that insomnia risks increase with age.1,3,4,6 Additionally, sleep needs change over the life span,31 with infants requiring the most sleep and adults requiring the least. With Mary, it was important to question whether she was experiencing insomnia or whether she just required less sleep as she got older. If done properly, sleep restriction and sleep consolidation should reveal a person’s current sleep needs by slowly changing a person’s amount of time in bed based on their sleep efficiency.
Work Status
As someone who was not working, Mary was at a higher risk for developing insomnia.3,4,6 She presented with a lot of unstructured time at home during the day, which had historically allowed her to nap. Fortunately, she did have a regular wake-up time due to needing to drive her daughter to school, which helped facilitate the structured wake-up time in the sleep restriction section of the intervention.
Physical Activity Level
Mary is a very active individual, walking daily even through her active cancer treatment. The hope was that her physical activity would help drive her sleep debt and increase her ability to fall asleep at night.
Limitations
Lack of Interest in Doing Sleep Log
Mary’s strong desire not to complete sleep logs resulted in acknowledgment in a session that there may not be accurate information about the amount of time she actually spent in bed and the amount of time she actually slept, thus only being able to estimate her sleep efficiency and taking precision out of the sleep restriction technique.
Lack of Desire to Stop Certain Sleep Habits
Mary was set in her ways about certain habits and was adamant that there were certain things she did not want to give up, including reading, watching TV, and using her tablet in bed. Additionally, she felt that the stimulus control technique of getting out of bed when not sleeping felt odd and was not a behavior that she planned on engaging in. Subsequently, it was important to consider other techniques that might help her with her sleep despite not wanting to engage in these particular behaviors.
Place/Frequency of Clinical Encounters
Mary lived far enough away from the cancer center that it was not convenient for her to attend sessions weekly. This factored into the treatment related to starting her on an intervention the first day. Traditionally, the first full session would have been used for the assessment, and the patient would have been assigned homework of sleep log completion before the next session. The quick-start nature of this intervention resulted in the first session being heavily intervention focused, something common to a health setting but not a traditional mental health setting. Because Mary was not going to come in weekly, beginning the intervention right away helped build rapport, get patient buy-in, and provided her with techniques to make her feel successful as quickly as possible.
Implications and Recommendations
Above is the description of a CBT intervention for sleep with an individual at a medical center. There were many individual considerations that were pertinent to the case and the CBT work with her. Although not a traditional mental health setting with regard to frequency or number of visits, the CBT intervention was successful for Mary, as she was able to note changes in her sleep and anxiety after the first visit when she started making behavioral and cognitive changes. While she did not buy into all of the techniques suggested, she was able to make significant changes, which, through self-report, suggest improved sleep and anxiety.
Ideally, Mary would have engaged in treatment for a longer time to make sure that her sleep and anxiety were sustained, and to continue with additional cognitive and behavioral techniques that may have led to better sleep. Beyond self-report, there are no measures of anxiety that were provided at the end of treatment to be able to compare objectively the changes in anxiety over the course of treatment. Similarly, no objective measures of sleep were completed (either pretreatment or posttreatment), so all information is subjective. That being said, her subjective feeling was that her anxiety and sleep improved over the course of the treatment. Hopefully Mary’s anxiety and sleep will continue to improve as she persists using the techniques she employed to help maintain good sleep.
Sleep difficulties are common for people who have been diagnosed with cancer. Research suggests that no one is immune from developing insomnia, and sleep issues are at a high rate in the United States. However, health disparities exist; research suggests that far fewer Asians (28%), as opposed to other ethnic groups (48% of whites, 42% of blacks, and 40% of Hispanics), have been asked about sleep by their doctor.5 This health disparity could be prevented by universal screening about sleep issues at every healthcare encounter. Early detection could be helpful to patients to improve their quality of life. As with the case of Mary, small, quick interventions for sleep, which could easily be delivered in a healthcare setting, could reduce suffering and even save lives.
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition: DSM-5. Washington, DC; 2013.
- Ohayon MM. Prevalence and comorbidity of sleep disorders in general population. Rev Prat. 2007;57:1521-1528.
- Bixler EO, Kales A, Soldatos CR. Sleep disorders encountered in medical practice: a national survey of physicians. Behav Med. 1979;6:13-21.
- Mellinger GD, Balter MB, Uhlenhuth EH. Insomnia and its treatment. Prevalence and correlates. Arch Gen Psychiatry. 1985;42:225-232.
- National Sleep Foundation. 2010 Sleep in America poll. Sleep and Ethnicity. https://sleepfoundation.org/sleep-polls-data/sleep-in-america-poll/2010-sleep-and-ethnicity. Published 2010. Accessed January 30, 2015.
- Ohayon MM, Caulet M, Priest RG, et al. DSM-IV and ICSD-90 insomnia symptoms and sleep dissatisfaction. Brit J Psychiatry. 1997;171:382-388.
- Roscoe JA, Kaufman ME, Matteson-Rusby SE, et al. Cancer-related fatigue and sleep disorders. Oncologist. 2007;12:35-42.
- Vena C, Parker K, Cunningham M, et al. Sleep-wake disturbances in people with cancer part I: an overview of sleep, sleep regulation, and effects of disease and treatment. Oncol Nurs Forum. 2004;31:735-746.
- Kuppermann M, Lubeck DP, Mazonson PD, et al. Sleep problems and their correlates in a working population. J Gen Intern Med. 1995;10:25-32.
- Simon GE, VonKorff M. Prevalence, burden, and treatment of insomnia in primary care. Am J Psychiatry. 1997;154:1417-1423.
- Berger AM. Update on the state of the science: sleep-wake disturbances in adult patients with cancer. Oncol Nurs Forum. 2009;36:E165-E177.
- Ebben MR, Spielman AJ. Non-pharmacological treatments for insomnia. J Behav Med. 2009;32:244-254.
- Irwin MR, Cole JC, Nicassio PM. Comparative meta-analysis of behavioral interventions for insomnia and their efficacy in middle-aged adults and in older adults 55+ years of age. Health Psychol. 2006;25:3-14.
- Mitchell MD, Gehrman P, Perlis M, et al. Comparative effectiveness of cognitive behavioral therapy for insomnia: a systematic review. BMC Fam Pract. 2012;13:40.
- Montgomery P, Dennis J. Cognitive-behavioral interventions for sleep problems in adults aged 60+. Cochrane Database Syst Rev. 2002:CD003161.
- Morin CM, Culbert JP, Schwartz SM. Nonpharmacological interventions for insomnia: a meta-analysis of treatment efficacy. Am J Psychiatry. 1994;151:1172-1180.
- Murtagh DR, Greenwood KM. Identifying effective psychological treatments for insomnia: a meta-analysis. J Consult Clin Psychol. 1995; 63:79-89.
- Okajima I, Komada Y, Inoue Y. A meta-analysis on the treatment effectiveness of cognitive behavioral therapy for primary insomnia. Sleep Biol Rhythms. 2011;9:24-34.
- Hauri PJ. Sleep Hygiene, Relaxation Therapy, and Cognitive Interventions. In: Hauri PJ, ed. Critical Issues in Psychiatry: Case Studies in Insomnia. New York, NY: Plenum Medical Book Co; 1991:65-84.
- Morgenthaler T, Kramer M, Alessi C, et al. Practice parameters for the psychological and behavioral treatment of insomnia: an update. An American Academy of Sleep Medicine report. Sleep. 2006;29:1415-1419.
- Clark J, Cunningham M, McMillan S, et al. Sleep-wake disturbances in people with cancer part II: evaluating the evidence for clinical decision making. Oncol Nurs Forum. 2004;31:747-771.
- Palesh OG, Roscoe JA, Mustian KM, et al. Prevalence, demographics, and psychological association of sleep disruption in patients with cancer: University of Rochester Cancer Center–Community Clinical Oncology Program. J Clin Onc. 2010;28:292-298.
- Phillips KM, Jim HS, Donovan KA, et al. Characteristics and correlates of sleep disturbances in cancer patients. Support Care Cancer. 2012;20:357-365.
- Savard J, Morin CM. Insomnia in the context of cancer: a review of a neglected problem. J Clin Oncol. 2001;19:895-908.
- Johnson JA, Rash JA, Campbell TS, et al. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for insomnia (CBT-I) in cancer survivors. Sleep Med Rev. 2016;27:20-28.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Stewart DE, Cheung AM, Duff S, et al. Attributions of cause and recurrence in long-term breast cancer survivors. Psychooncology. 2001;10;179-183.
- Desai K, Mao JJ, Su I, et al. Prevalence and risk factors for insomnia among breast cancer patients on aromatase inhibitors. Support Care Cancer. 2013;21:43-51.
- Krishnan V, Collop NA. Gender differences in sleep disorders. Curr Opin Pulm Med. 2006;12:383-389.
- National Sleep Foundation. 2002 “Sleep in America” Poll. https://sleepfoundation.org/sites/default/files/2002SleepInAmericaPoll.pdf. Published 2002. Accessed May 17, 2016.




